MAURICE BRODIE, M.D. AND WILLIAM H. PARK, M.D., NEW YORK
Journal of the American Medical Association 105 no. 14 (Oct. 5, 1935): 1089-1093
Since individuals who have recovered from one attack of poliomyelitis rarely if ever are subject to a second one, there appears to be some basis for the belief that active immunization can be effective in this disease. Moreover, both convalescent human beings and monkeys usually have demonstrable antibody.
We have found the antibody content higher following a severe attack as compared with that produced by a mild attack in the experimental animal, and likewise animals recovered from a severe attack show a much greater resistance to reinfection. Since there is some correlation between concentration of antibody and immunity, the former can be used as an index of immunity in immunization experiments.
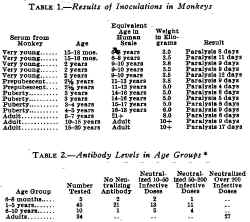
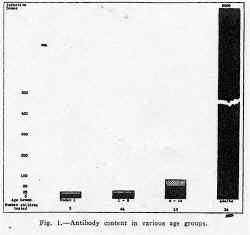
Epidemiologic evidence indicates that immunity against poliomyelitis develops with aging and is probably due to exposure to the virus. Added evidence of this is brought out by a study of antibody in the serums of human beings and monkeys at different age levels. In monkeys, owing to a lack of exposure to the virus (table 1), there is no antibody at any age, but in human beings (table 2 and fig. 1) there is increased antibody with aging. Since in natural immunization some children contract the disease, a safe and effective method of artificial immunization would be preferable. For this purpose, only that vaccine which is antigenic, safe and innocuous would be of any use. This report deals with the immunization of human beings and monkeys with such a vaccine and with the tests performed to determine the degree of this immunity.
Toward this end, several methods were first developed in monkeys. The first involved the injection of subinfective doses of active virus. This was discarded because of the danger of infection. The second method, which was concerned with the use of virus serum mixtures, was not practical because of the fact that in order to get an appreciable immunity the mixtures had to be just neutralized, which meant the use of many monkeys. The third and selected preparation was that of virus inactivated by germicides.
Infectious monkey cord was treated with various germicides, of which solution of formaldehyde proved to be the most efficacious. Various dilutions of this germicide were mixed with the virus and the mixture was kept either in the incubator or in the icebox for different intervals, to inactivate the virus. In each case equal parts of double the required concentration of solution of formaldehyde and 20 per cent virus suspension were combined. During the course of this work it was found that the virus suspension should be just inactivated, for overtreatment or prolonged treatment with solution of formaldehyde reduced the antigenicity of the vaccine (7). Therefore, in most of the work reported, virus inactivated by 0.1 per cent solution of formaldehyde for from eight to twelve hours at 37 C. was usually employed.
In immunizing the experimental animal, the vaccine was administered in either one or two doses of 2.5, 5 or 10 cc. amounts. It was found that a 5 cc. dose was sufficiently antigenic so as to produce an immunity better than that developed after a mild attack of the disease and sometimes comparable with that produced by either immunization with active virus or a severe attack of poliomyelitis.
Experiments in monkeys indicated that immunity could be developed by the injection of virus treated with solution of formaldehyde and that the vaccine was noninfective (table 3). Inoculation of this material into several human volunteers having shown that it was probably safe for human administration, it was used in children.
The vaccine was given in either one or two doses, each 5 cc. in amount, of which from 1 to 2 cc. was injected intracutaneously and the remainder subcutaneously. In more than 2,300 vaccinations there have been only three general reactions and twenty-three appreciable local reactions, such as superficial necrosis or marked induration. About one third of those vaccinated showed a moderate induration, which lasted for a week or two.
 About seventy-five children have been tested for antibody both
before and after immunization. The results are herewith recorded so far
as they have been completed. To test for antibody, the serums both before
and for four weeks after the administration of the vaccine were mixed
with varying amounts of virus; the mixtures were incubated at 37 C. for
two hours and after standing on ice over night, were injected into monkeys,
intracerebrally. If neutralization occurred, the monkey remained well.
The test can be carried out quantitatively, and so, in those children
in whom a slight amount of antibody was present, it was possible to determine
whether or not an increase occurred after immunization.
About seventy-five children have been tested for antibody both
before and after immunization. The results are herewith recorded so far
as they have been completed. To test for antibody, the serums both before
and for four weeks after the administration of the vaccine were mixed
with varying amounts of virus; the mixtures were incubated at 37 C. for
two hours and after standing on ice over night, were injected into monkeys,
intracerebrally. If neutralization occurred, the monkey remained well.
The test can be carried out quantitatively, and so, in those children
in whom a slight amount of antibody was present, it was possible to determine
whether or not an increase occurred after immunization.
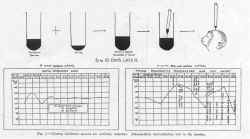
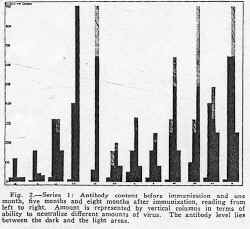
The first twelve children, of whom five received single 5 cc. dose and seven, two such doses, eleven days apart, were vaccinated in July 1934. They were tested before and at one month, five months and eight months after immunization. When tested from three to five weeks later, all showed an appreciable antibody response. At five months, nine of eleven retained a considerable amount of antibody, while at eight months, seven of the nine still showed antibody. These results are summarized in figure 2.
A second series of seventeen children was then vaccinated, of which seven received 5 cc. amounts, four a single dose, and three two such doses, while ten were given 2.5 cc. amounts, five a single dose and five two doses. The methods of immunization and the test carried out before and at one and five months after immunization are given in table 4, in which it can be seen that the immunity resulting from the injection of 5 cc. amounts is decidedly better than with 2.5 cc amounts.
 The
results up to the present show that, with a single 5 cc. dose, antibody
was produced in twenty-two of twenty-six children and that with two such
doses ten out of ten responded. With one dose of 2.5 cc., four out of
five developed antibody and with two such doses five out of five showed
an antibody response. That two doses each of 5 cc. are decidedly better
than a single dose or than 2.5 cc. amounts is indicated in figure 3, in
which it is shown that the antibody is better maintained with two doses
than with one or with the smaller amount of vaccine. One child, who before
immunization had no antibody and who, after a single dose, showed no response,
was then given a second dose, after which there was demonstrable antibody
in his serum. Two children, who had received a single dose, lost their
antibody at five and eight months respectively and were given a second
dose. After the reinjection of the vaccine a rapid and high antibody response
followed. It would seem, therefore, that two doses rather than one should
be given.
The
results up to the present show that, with a single 5 cc. dose, antibody
was produced in twenty-two of twenty-six children and that with two such
doses ten out of ten responded. With one dose of 2.5 cc., four out of
five developed antibody and with two such doses five out of five showed
an antibody response. That two doses each of 5 cc. are decidedly better
than a single dose or than 2.5 cc. amounts is indicated in figure 3, in
which it is shown that the antibody is better maintained with two doses
than with one or with the smaller amount of vaccine. One child, who before
immunization had no antibody and who, after a single dose, showed no response,
was then given a second dose, after which there was demonstrable antibody
in his serum. Two children, who had received a single dose, lost their
antibody at five and eight months respectively and were given a second
dose. After the reinjection of the vaccine a rapid and high antibody response
followed. It would seem, therefore, that two doses rather than one should
be given.
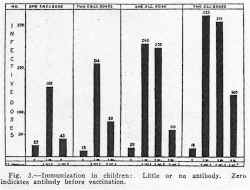
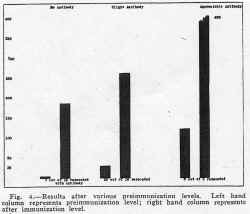
The serums of the majority of the children immunized had little or no antibody before immunization, while a few had considerable neutralizing power. For the purpose of study they have been divided into three groups: those without any antibody, those with a slight amount, and those with appreciable antibodies. The first of these groups also includes serums neutralizing less than ten infective doses, the second from ten to fifty and the third more than fifty infective doses. The results obtained from thirty-three children given 5 cc. amounts of vaccine are represented in figure 4, in which it is shown that although the proportion of those immunized and the height of antibody level is greater when antibody is present before vaccination, yet in the absence of any previous antibody in the small series, nine out of twelve developed antibody. All but two of these were given a single dose and since a second dose may produce antibody, it is quite likely that, judging from the small series in which one dose gave immunity to 75 per cent, two doses would produce antibody in a greater proportion. Thus the vaccine appears to elicit an antibody response in those who seem to be the most susceptible, in that they have no normal antibody.
 For
use in epidemics it is important that the administration should be followed
by the rapid production of immunity. Three children who had small amounts
of antibody before vaccination were tested at weekly intervals after receiving
the vaccine. Demonstrable antibody was present in a week and reached its
height in three to four weeks. In two children, who had no antiviral substance
in the control period, antibody could be demonstrated in eight days in
one and in the other at sixteen days following vaccination. Figure 5 shows
the composite antibody production of three children tested at weekly intervals
for four weeks and then at six and eight weeks after injection of the
vaccine. In the same graph is indicated the amount of antibody present
at various intervals following the stage of acute paralysis in three convalescent
cases. The results show that the degree of immunity developed by vaccination
compares favorably with that present in the three children who recovered.
For
use in epidemics it is important that the administration should be followed
by the rapid production of immunity. Three children who had small amounts
of antibody before vaccination were tested at weekly intervals after receiving
the vaccine. Demonstrable antibody was present in a week and reached its
height in three to four weeks. In two children, who had no antiviral substance
in the control period, antibody could be demonstrated in eight days in
one and in the other at sixteen days following vaccination. Figure 5 shows
the composite antibody production of three children tested at weekly intervals
for four weeks and then at six and eight weeks after injection of the
vaccine. In the same graph is indicated the amount of antibody present
at various intervals following the stage of acute paralysis in three convalescent
cases. The results show that the degree of immunity developed by vaccination
compares favorably with that present in the three children who recovered.
 Were
a simple test for antibody available to enable the selection of those
requiring the vaccine and also to determine the results of immunization,
it would be possible to limit the number of vaccinations. The only test
available at the present time is the monkey neutralization test, which
involves the use of several monkeys and also an interval of from two to
four weeks. We were able to render mice susceptible to the virus of poliomyelitis
by subjecting them to repeated x-ray exposures and to transfer the infectious
agent serially to normal mice. The fact that we were transferring the
virus of poliomyelitis was borne out by specific neutralization of the
mouse passage virus by serums containing polio- myelitis antibody, by
the production of typical poliomye- litis in monkeys who were injected
with infectious mouse brains, and by the immunization of monkeys with
the mouse virus. However, after a number of passages the virus is lost
in mice and has to be reestablished.
Were
a simple test for antibody available to enable the selection of those
requiring the vaccine and also to determine the results of immunization,
it would be possible to limit the number of vaccinations. The only test
available at the present time is the monkey neutralization test, which
involves the use of several monkeys and also an interval of from two to
four weeks. We were able to render mice susceptible to the virus of poliomyelitis
by subjecting them to repeated x-ray exposures and to transfer the infectious
agent serially to normal mice. The fact that we were transferring the
virus of poliomyelitis was borne out by specific neutralization of the
mouse passage virus by serums containing polio- myelitis antibody, by
the production of typical poliomye- litis in monkeys who were injected
with infectious mouse brains, and by the immunization of monkeys with
the mouse virus. However, after a number of passages the virus is lost
in mice and has to be reestablished.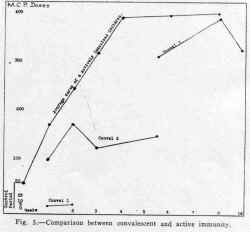
Although in children the vaccine produces an antibody response which compared well with that of several convelescent children, the test of its value must rest with epidemic studies and with a follow up of the incidence of the disease in a large number of grouped, vaccinated children. Although the vaccine was used in a small outbreak in Kern County, Calif., the incidence of the disease was too low and the number vaccinated too few, as indicated in Table 5, to give any definite information. Large numbers of children are being immunized in several centers in New York and in Newark, N. J., and are being followed (fig. 6) with controls. We hope to immunize a sufficient number to make a comparative study between the immunized and the nonimmunized.
 A
poliomyelitis vaccine treated with formaldehyde gives antibody production,
which is apparent in about one week and reaches its full development in
from three to four weeks. It has been demonstrated eight months after
vaccination and by greater amounts in those receiving two doses. As far
as the tests have been carried out, a single dose produces antiviral substance
in more than 75 per cent and two doses in practically all.
A
poliomyelitis vaccine treated with formaldehyde gives antibody production,
which is apparent in about one week and reaches its full development in
from three to four weeks. It has been demonstrated eight months after
vaccination and by greater amounts in those receiving two doses. As far
as the tests have been carried out, a single dose produces antiviral substance
in more than 75 per cent and two doses in practically all.
 The
material appears to be perfectly safe, as demonstrated by its noninfectivity
for monkeys and from more than 2,300 human immunizations.
The
material appears to be perfectly safe, as demonstrated by its noninfectivity
for monkeys and from more than 2,300 human immunizations.
ABSTRACT OF DISCUSSION
DR. JOHN A. KOLMER, Philadelphia: Three years ago my associates and I failed to immunize monkeys with vaccines of poliomyelitis virus apparently "killed" with heat, phenol, chloroform and other chemical agents and turned our attention to immunization with living virus attenuated with sodium ricinoleate, which proved completely safe and apparently highly effective. It may be that our failure with "dead" virus was due to the administration of too small doses, but I am convinced that the living attenuated virus in small doses is a safe and effective immunizing agent. None of a large group of monkeys receiving as many as ten consecutive subcutaneous injections of the ricinoleated vaccine in doses varying from 0.05 to 1 cc, per kilogram of weight showed the slightest evidences of infection during the period of immunization and all were found to have acquired a high degree of immunity as determined by the intracerebral injection of virus. Sodium ricinoleate vaccine has now been given to more than 400 children in Philadelphia during the past year with absolutely no ill effects, and enough for the immunization of more than2,000 individuals has been distributed. No effects other than mild local reactions at have occurred. Tbe safety of the vaccine is largely due to the fact that it is prepared from remote monkey passage virus that has apparently lost infectivity for human beings, just as the smallpox virus is changed by passage through the lower animals. Additional factors of safety are the subcutaneous route of administration, the use of three small doses at weekly intervals and the rapidity of antibody production. Antibody has been found in the blood of children as early as ninety-six hours after the first dose, and during the past year we have found that monkeys can be effectively protected when the vaccine is given even during the incubation period of the disease following intracerebral injections of virus. For this reason I believe that the vaccine can be given safely during epidemics and during the incubation period. Attenuation of the virus with sodium ricinoleate may be an additional factor of safety, but the degree of attenuation is slight and of minor importance. Antibody production occurs in at least 80 to 90 per cent of vaccinated children with the doses now employed, comparable in amount to that found in the blood of individuals recovering from an attack of the disease. I too believe that this antibody will protect against the disease and we have found that it was capable of neutralizing the virus of the California epidemic.
DR, ALTON GOLDBLOOM, Montreal: It is gratifying to see the extent to which Drs. Brodie and Park have succeeded in producing a harmless inoculation capable of producing immunity against poliomyelitis on a large scale. One or two practical points have to be considered in the application of this immunization It must be realized that it can never be either necessary or possible to immunize the entire child population of so large a country as this against poliomyelitis. It would be desirable to have some easy, rapid method of determining those children who are susceptible to poliomyelitis and to use the methods available for them. A rapid test, therefore, is essential as one of the prerequisites of this method. In some bacterial diseases susceptibility can be determined by skin tests. Those methods are scarcely available in the case of virus diseases, but perhaps some new approach to the entire subject of immunity to virus diseases may eventually develop a new index of susceptibility or immunity, and with that may come an easy, rapid method of separating susceptible from nonsusceptible subjects. In addition, it is essential to develop an easier and cheaper means of producing virus for the purpose of formalizing. At present virus is produced only through the destruction of infected monkeys The authors have indicated the possibility of developing virus from some cheap, easily handled and easily available animal, such as the mouse. They have shown that the exposure of mice to x-rays renders them susceptible to the infection and it may be that some such method will be developed by which virus will be available on a large scale; or some rnethod of culture of the virus itself, either by tissue methods or some other new medium, will be developed which will make the culture of viruses as simple a thing as the culture of bacteria is today. Again, this may conic about, as Dr. Kolmer indicated, by the recognition of one of the many paralyzing diseases of animals as being so allied to poliomyelitis that it may in itself be antigenic against poliomyelitis in human beings, without being pathogenic.
DR. MAURICE BRODIE, New York: I agree with Dr. Kolmer when he states that decidedly larger amounts of an inactivated virus or vaccine are necessary. Of course, that has been pointed out in other diseases, in which it has been shown that inactivated virus will immunize as long as large amounts are used. With active virus one can use smaller amounts. However, we found that with the use of very small amounts of virus, such as would be safe from danger of infection, we had to give repeated doses to immunize. To get immunization with one or two doses, it was necessary to give a fairly large dose, about half to one fourth as much as of formalized vaccine. That dose was dangerous for monkeys and occasionally some came down with mild or even severe attacks of the disease. In order to get immunization with the very small and safe doses, we had to give many doses, which is not practical, and so it seemed more practical to use two larger doses of formalized vaccine. Although I think the small doses of the active virus may he perfectly safe, it has to have a very widespread use to determine that. I do not feel we can say the virus has been changed as Dr. Kolmer has stated. In rabies, for example, the rabbit virus is still under discussion as to whether the paralytic accidents of rabies immunization are due to rabies virus or not. And so, since, in rabies, immunization has been going on many Iyears and still the question is not answered, in poliomyelitis, whether or not active virus will infect human beings can be answered only when thousands have been immunized and another susceptible animal is found. If it were possible now to infect mice, the virus could be passed through hundreds of generations and it would be seen whether or not it still infected monkeys. Dr. Goldbloom has pointed out the impracticability of the immunization except in an epidemic.
Copyright © 1997 University
of Pennsylvania HSS
Revised: